>> Research
| Publications
| Google Scholar
| Pubmed
| ORCID
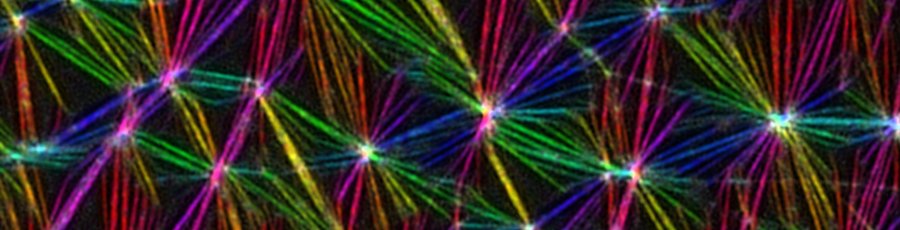
-> NEW ARTICLE New paper from our lab in collaboration with Richard Morris at UNSW and EMBL Australia!
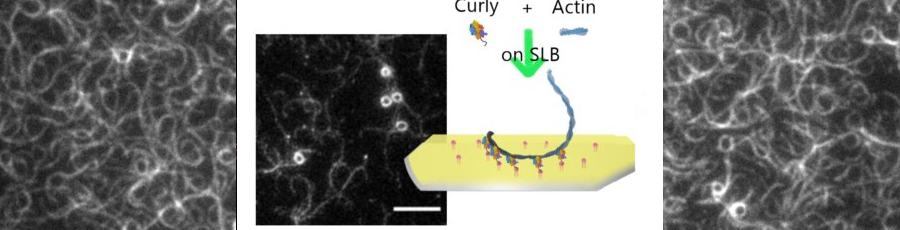
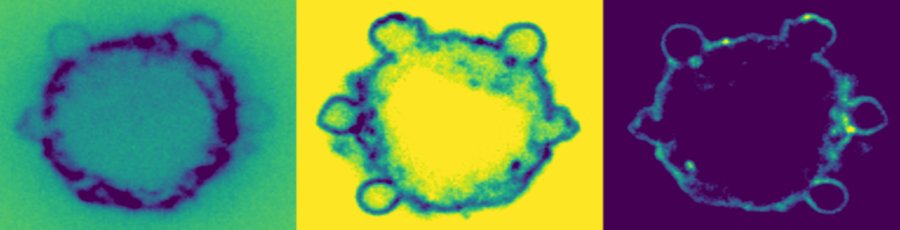
-> PREPRINT New preprint based on the work of Sayantika Ghosh on our fascinating observations of actin organisation in cells during E-cadherin driven adhesion!
 Darius Köster (group leader, contact, @ActinDomain, LinkedIn) |CV
Darius Köster (group leader, contact, @ActinDomain, LinkedIn) |CV
How do cells cope with their environment? Deciphering mechanical processes at the cell surface leading to signalling events and the adaptation of cells to changes in the environment.
My research interests lie in the understanding the molecular and physical principles that govern processes at the plasma membrane of cells. Particularly, by which mechanisms the force generating machinery of the cell cortex,
structural filaments and motor proteins, govern and regulate the mechanical properties of the cell membrane and dynamics of cell membrane components, and vice versa, how membrane organisation and signalling events feed-back
to the regulation of the cortex machinery. These mechanisms, which in turn regulate cell motility and cell-cell interactions, underlie important, poorly-understood human diseases that constitute global health problems.
The lab employs novel assays based on reconstituted membrane systems in combination with measurements on live cells using state of the art fluorescence microscopy and mechanical manipulation.
 John James (funded by EPSRC, LinkedIn)
John James (funded by EPSRC, LinkedIn)
I am a post-doctoral researcher with extended experience in cell biology, genetic engineering and microscopy, supplemented by basic knowledge of biochemistry and computational data analysis. I utilise this skill-set to study the interplay between actin-generated forces and the maturation of cell adhesions.
During my PhD at the lab of Alexis Gautreau (Ecole Polytechnique, France), I showed that vinculin, a mechano-sensor present at focal adhesions and adherens junctions, suppresses the generation of branched actin networks. This signal impacts several cell behaviours ranging from proliferation to migration to attachment, potentially establishing a novel tumor-suppressor pathway. I now join the Koester Lab with the hope of understanding how forces generated by these branched actin networks can in turn influence the maturation and stability of cell adhesions. Uncovering such an inverse regulation could complete a feedback loop essential for the sustenance of cell adhesion and thus the prevention of cancer metastasis.
 Sedigheh (Mobi) Ghanbarzadeh Nodehi (funded by EPSRC)
Sedigheh (Mobi) Ghanbarzadeh Nodehi (funded by EPSRC)
I moved from a physics background to discovering the underlying principles in cell biology. Like every physicist does, I try to simplify the multilayered complexity in biology through a minimal set of components that can sufficiently model and describe biological phenomena. The most intriguing question in biology for me is how cells understand their mechanical environments and convey these signals to the nuclei. Having this perspective, I did my Ph.D. in studying the mechanical characteristics of the nuclear membrane of stem cells under Dr. Farshid Mohammad-Rafiee (IASBS Zanjan) and in collaboration with Prof. Jacques Prost (Institut Curie) and Prof. G.V. Shivashankar (ETH Zurich). I joined the Koester lab as a postdoctoral fellow at the University of Warwick through an EPSRC-funded project to dig into actin dynamics. I aim to study how the actomyosin cortex deforms lipid membranes in a minimal system and how actin length and curvature distributions might play a role in mediating biological processes.
 Lea Denker (completed PhD in Dec 2024, MIBTP)
Lea Denker (completed PhD in Dec 2024, MIBTP)
I am interested in studying the coronavirus replication mechanism. Specifically, my research focusses on the non-structural proteins nsp3, nsp4, and nsp6, which have previously been shown to be vital for the formation of a viral replication platform. Yet, the extent of their involvement and specific functions remains to be elucidated. Therefore, the aim is to explore the interaction of these non-structural proteins with synthetic lipid membranes to determine their role in membrane binding and membrane deformation. This can be achieved by using giant unilamellar vesicles and optical fluorescence microscopy techniques.
 Paulina Pokorska (completed PhD in Jan 2025, ARAP program, with Aparna Ratheesh at CMCB and Walter Hunziker at IMCB Singapore, @PaulinaPokorsk2)
Paulina Pokorska (completed PhD in Jan 2025, ARAP program, with Aparna Ratheesh at CMCB and Walter Hunziker at IMCB Singapore, @PaulinaPokorsk2)
The main focus of my PhD project is on cell-cell adhesions, in particular septate junctions. Septate junctions are cellular junctions present in Drosophila melanogaster, which are known to be homologues to tight junctions in mammalian cells and are essential to maintain cell polarity and required for the proper functioning of epithelial tissue.
My PhD project is focused on studying spatial and temporal organization of septate junctions throughout embryonic development. This will be achieved in collaboration with my supervisor from Singapore, Dr Walter Hunziker. In Singapore, I will use APEX2 mediated proximity biotinylation and electron microscopy combined with proteomics studies. I will also characterize fine tuning role of extracellular matrix deposited by macrophages and tissue mechanics in the formation of these junctions. The success of this project will result in major advances in our understanding of the dynamic regulation of cell-cell adhesions and molecular details of a process integral for fly development.
 Mary Fesenko (supported by career development funded of MRC-DTP)
Mary Fesenko (supported by career development funded of MRC-DTP)
---PhD Candidates---
 Lucie Winn (MRC-DTP)
Lucie Winn (MRC-DTP)
My PhD project focusses on cell-cell adhesion, particularly via E-cadherin. Cadherins can recruit adapter proteins which link the cell-cell adhesion site to the actomyosin cytoskeleton of cells. This is a mechanosensitive process, and factors such as E-cadherin mobility impact regulation of the cytoskeleton. However, the key mechanisms and cascades of such mechanosensitive signalling remain unknown. During my PhD I hope to gain a better understanding of this process, and delve into questions such as, is mobility-mediated regulation local (at contact site only) or global (translated over the entire cell surface)? How are different proteins activated to organise the cytoskeleton? And how do other mechanical constraints affect E-cadherin mechano-signalling? We hope a better understanding of this fundamental biological process will provide important insights into several other related biological process, such as tissue development, wound healing and cancer metastasis.
 Badeer Hassan Ummad (Chancellor International Scholarship, LinkedIn, @BadeerHassan)
Badeer Hassan Ummad (Chancellor International Scholarship, LinkedIn, @BadeerHassan)
My research interest lies in integrating quantitative techniques in biology to understand complex cellular phenomena. I started exploring biology during my master at IISER Pune by studying cytoskeletal organisation inside cellular geometry using computational modelling approaches. These experiences got me fascinated with the applications of quantitative methods to study biology. At Koester lab, I will be looking into the interaction of actin cytoskeleton with cellular membrane. This research will help to learn how the cytoskeletal network organisation determines cell shape and how biochemical and mechanical signals control this process.
 Sabeeha Malek (MIBTP, @SabeehaMalek)
Sabeeha Malek (MIBTP, @SabeehaMalek)
I am interested in gaining a deeper understanding of cell adhesion and cell biomechanics in the Ehlers-Danlos Syndromes (EDS) and Hypermobility Spectrum Disorders (HSD). It has been established that these conditions demonstrate an altered integrin profile with a defective extracellular matrix, yet the consequences of this on the biomechanical properties of EDS/HSD fibroblasts has not been explored. I wish to use live cell microscopic techniques in combination with the mechanical manipulation of single cells to gain a better understanding of the dynamic processes involved in maintaining tissue integrity.
 Peter Mottram-Epson
Peter Mottram-Epson
I am a Medical Sciences MRes student , who is looking into mammalian epithelial cell to cell adhesion dynamics with a supported lipid bilayer under confinement, using TIRF microscopy.
----------------Former lab members----------------
Sayantika Ghosh (completed PhD in Feb 2025, Chancellor International Scholarship)
Thesis title: To study the E-cadherin-based junction formation in real-time in cancer cells using a minimal system.
project students
- Alina Makhmudova (visiting from University of Geneva)
- Deepak Ishwara Shivaprakash (visiting from PES University Bangalore)
Post-docs
- Zoe Shofield (now at Birmingham University)
- Scott Clarke (now at WMG)
----------------------------------------------
A 1min description of our research...